
"Structure and function of the vacuolar H+-ATPase: moving from low-resolution models to high-resolution structures". ^ Jones R, Findlay JB, Harrison M, Durose L, Song CF, Barratt E, Trinick J (2003)."Cysteine-mediated cross-linking indicates that subunit C of the V-ATPase is in close proximity to subunits E and G of the V1 domain and subunit a of the V0 domain". 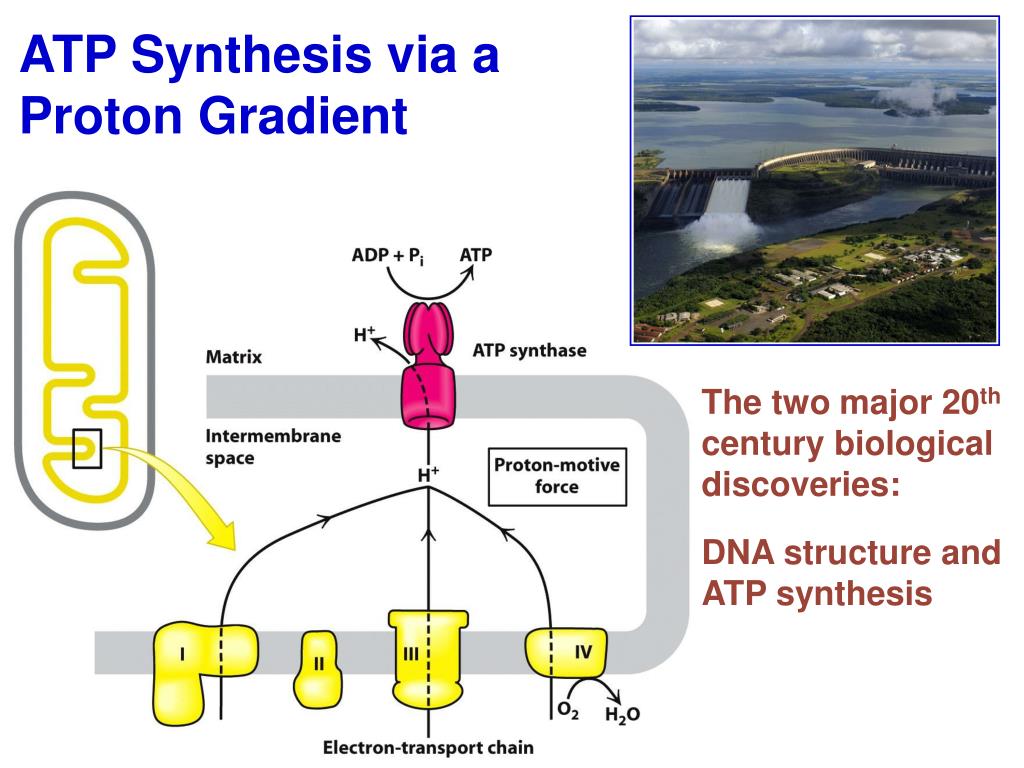
"Mechanics of coupling proton movements to c-ring rotation in ATP synthase". ^ Fillingame RH, Angevine CM, Dmitriev OY (2003)."A structural model of the vacuolar ATPase from transmission electron microscopy". "Resolution of distinct rotational substeps by submillisecond kinetic analysis of F1-ATPase". ^ Itoh H, Yoshida M, Yasuda R, Noji H, Kinosita K (2001)."Mechanisms of ATPases-a multi-disciplinary approach". "The evolution of A-, F-, and V-type ATP synthases and ATPases: reversals in function and changes in the H+/ATP coupling ratio". "Rotary ATPases: A New Twist to an Ancient Machine". ^ Kühlbrandt W, Davies KM (January 2016).Human proteins containing this domain ĪTP5G1 ATP5G2 ATP5G3 ATP6V0B ATP6V0C ATPase, Fo complex, subunit C InterPro: IPR000454.ATPase, Vo complex, proteolipid subunit C, InterPro: IPR000245. 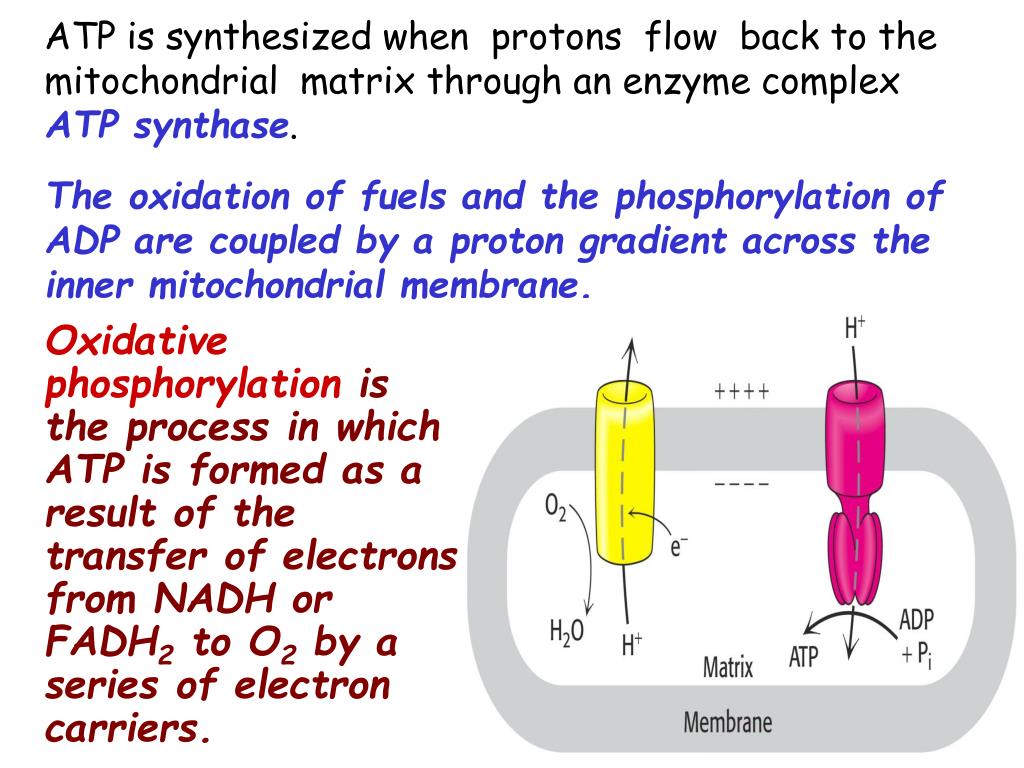
In a recent study c-subunit has been indicated as a critical component of the mitochondrial permeability transition pore. In V-ATPases, there are three proteolipid subunits (c, c′ and c′′) that form part of the proton-conducting pore, each containing a buried glutamic acid residue that is essential for proton transport. The sequential protonation and deprotonation of Asp61 of subunit C is coupled to the stepwise movement of the rotor. In F-ATPases, the flux of protons through the ATPase channel drives the rotation of the C subunit ring, which in turn is coupled to the rotation of the F1 complex gamma subunit rotor due to the permanent binding between the gamma and epsilon subunits of F1 and the C subunit ring of Fo. 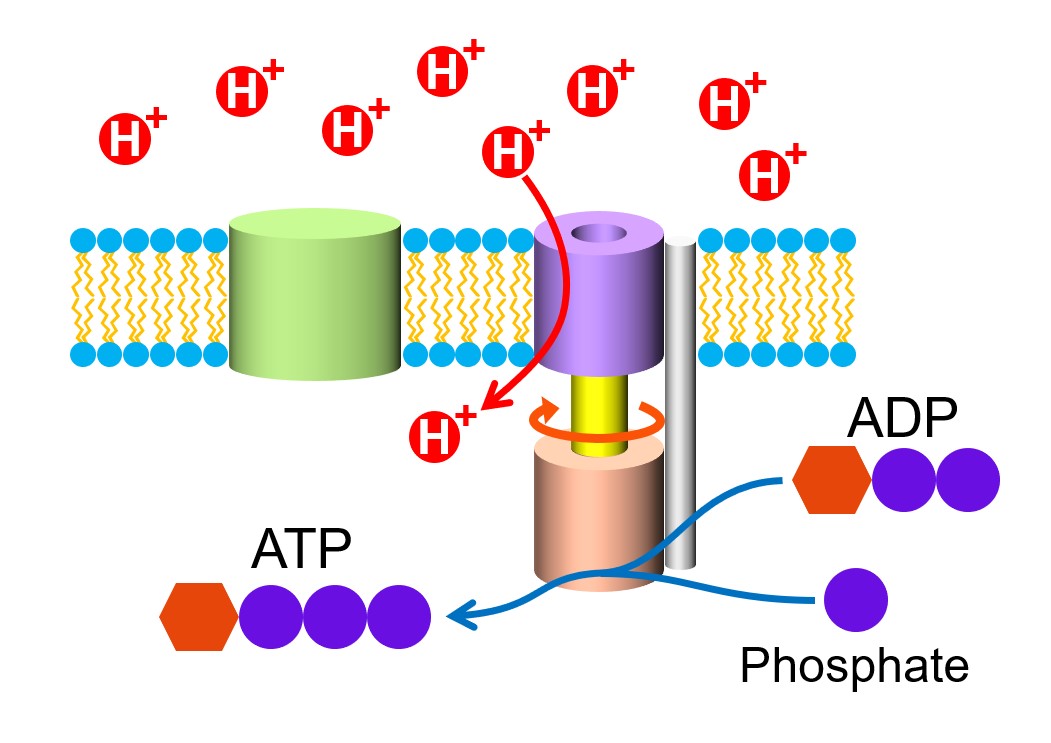
The F- and V-ATPases all contain rotary motors, one that drives proton translocation across the membrane and one that drives ATP synthesis/hydrolysis. The F-ATPases (or F1Fo ATPases) and V-ATPases (or V1Vo ATPases) are each composed of two linked complexes: the F1 or V1 complex contains the catalytic core that synthesizes/hydrolyses ATP, and the Fo or Vo complex that forms the membrane-spanning pore. There are different types of ATPases, which canĭiffer in function (ATP synthesis and/or hydrolysis), structure (F-, V- and A-ATPases contain rotary motors) and in the type of ions they transport. Using the energy from the hydrolysis of ATP to create a proton gradient. ATPases can harness the energy from a proton gradient, using the flux of ions across the membrane via the ATPase proton channel to drive the synthesis of ATP. ĪTPases (or ATP synthases) are membrane-bound enzyme complexes/ion transporters that combine ATP synthesis and/or hydrolysis with the transport of protons across a membrane. The subunits form an oligomeric c ring that make up the Fo/Vo/Ao rotor, where the actual number of subunits vary greatly among specific enzymes. Subunit C (also called subunit 9, or proteolipid in F-ATPases, or the 16 kDa proteolipid in V-ATPases) was found in the Fo or Vo complex of F- and V-ATPases, respectively. Calculated hydrocarbon boundaries of the lipid bilayer are shown by red and blue dotsĪTPase, subunit C of Fo/Vo complex is the main transmembrane subunit of V-type, A-type and F-type ATP synthases. V-type sodium ATPase from Enterococcus hirae.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
